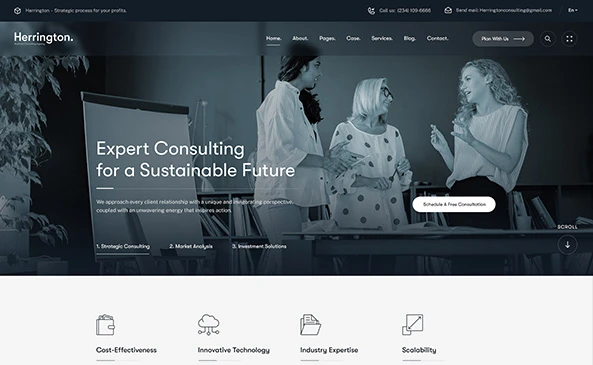
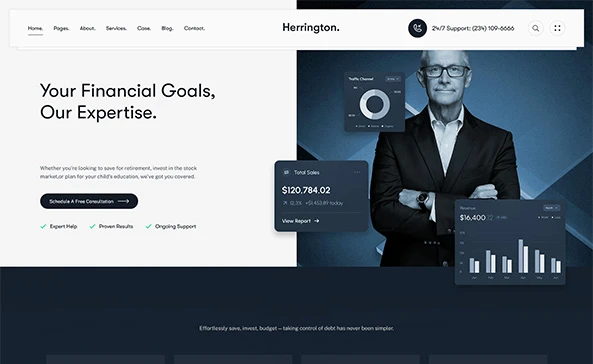
Developing a safe, effective, and secure MedTech product is not an accident.
We have cross-functional system expertise in sensors and catheters, mechanical and embedded design, software, and cloud technologies.
We can take a concept/ prototype through product development and production transfer, focusing on innovation and regulatory alignment.
You get a market-ready, compliant medical device product, fueled by our technical capabilities, MedTech domain knowledge and cost-effectiveness.

How we Help you Limit Risks and Launch your Business.
Through our experienced MedTech consultants, Vadanya Scientific helps you manage critical early-stage tasks with precision. By handling complex processes like compliance, design, and planning, we allow you to stay focused on your broader business goals without getting lost in the technical details.
We Develop MedTech Systems Spanning Following Technical Domains:
We provide the best services, ensuring your outstanding growth

Measurement System & Catheters

Product Design & 3D Modeling


Embedded Device Development

Windows/Mobile App Development


AI & Cloud-based Solutions
Product Development : Ongoing Projects

Device for early screening of an ophthalmology complication
• Currently in prototype development phase.
• Status: Clinical studies to improve image quality and design iterations
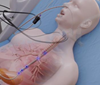
Smart Catheter for ICU Setting
• The next-gen design is being developed for an established medical device company
• Objective is to improve manufacturability, scalability and cost
• Status: Proof-of-concept verification
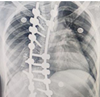
SAFER Spinal Fixation Surgery
• Status: Ideation and Planning


Why Vadanya Scientific ?
Seeking expert business advice early—before launching—is key. Vadanya Scientific work closely with you to identify the essential initial actions and guide your next moves.
Let’s Collaborate with Us!
13701 Marina Pointe Dr, #210
Call :+1-310-905-7597
Frequently Asked Questions
Some frequently asked questions about the service that you may have questions about
Who are your typical clients?
What is the process for beginning a project?
Can you help a client get funding?
What if my project lies outside your team’s skillset?
Will you tell me how to run my business?
Are you a data controller or processor?
Product Development : Past Experiences
Gastrointestinal functional disorders diagnostic systems
• System for diagnosing motility disorders in the
esophagus and lower GI tracts.
• Consists of a pressure-impedance sensing
catheter, data acquisition electronics, and
desktop-based software. .
Advanced Clinical Decision Support Software
• To enable physicians analyze complex clinical data, leading to patient diagnosis.
• Automated decision output to complement manual interpretation.
Mechanical design for a Continuous Glucose Monitoring (CGM) wearable
• Components in-scope: Needle-based skin implant,
Implant Insertion tool.
• Activities involved: Mechanical Modeling, Device Packaging, Process Development, and Clinical Validation.

Esophageal testing System for Endoscopy suite
• Simple user-friendly system for ad-hoc diagnosis within the complex endoscopy suite environment.

Portable Gastric Reflux Monitoring Device
• Multi-day monitoring of acid reflux to quantify gastric reflux, thereby guiding reflux treatment.