Scroll


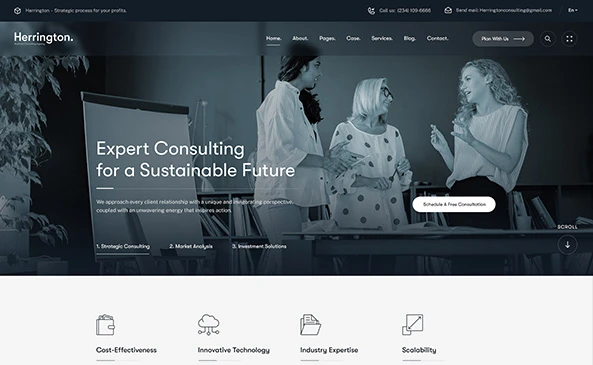
Welcome to Vadanya Scientific!
Vadanya Scientific is a boutique service and consultancy firm focused on development and commercialization of safe, effective and secure MedTech products. We understand that success in the medical technology space is never accidental—it’s the result of strategic planning, technical rigor, unwavering regulatory compliance and apt commercialization strategy.
We are a team of experienced professionals with deep expertise across the MedTech lifecycle. We believe that success comes from experience. Ironically, experience comes from bad experiences! Life is too short to make all the mistakes yourself: leverage our experience to achieve your success.
Within last 1.5 years
Our Vision
Deepening our cross-functional expertise
Achieving a high degree of operational excellence
Cultivating a culture of integrity, genuineness, and empathy within our team and beyond.
Following are some specific project categories we have recently worked upon :


Our MedTech Offerings
1. Product Development Service
1. Product Development Service
- Measurement System
- Product Design & 3D Modeling
- Embedded Device & Development
- Windows/Mobile App Development
- AI & Cloud Based Solution
3. Regulatory Compliance Consultancy
3. Regulatory Compliance Consultancy
- US FDA Indian CDSCO Submission
- ISO Standards Compliance
- Design History File
- Risk Assessment
- Safety Testing
2. Project and Process consultancy
2. Project and Process consultancy
- Usability Engineering
- Risk Management
- Design For Reliability
- Cyber Security
- Project Management
4. Merger & Acquisition Support
4. Merger & Acquisition Support
We bring you the domain expertise required to complement and collaborate with your M&A projects ,thus strengthening domain specific capabilities.
- Market Intelligence for US and India
- Technical due-diligence
- Post-merger Integration
Clinical Segments
Featured News and Insights
Read and stay updated with our latest news. We regularly share important updates and announcements.