Regulatory Compliance Consultancy
We have vast experience in the development and approval of complex medical devices and solutions for the US FDA, EU-MDR, and global regulatory bodies.
- Home Main
- Regulatory Compliance Consultancy

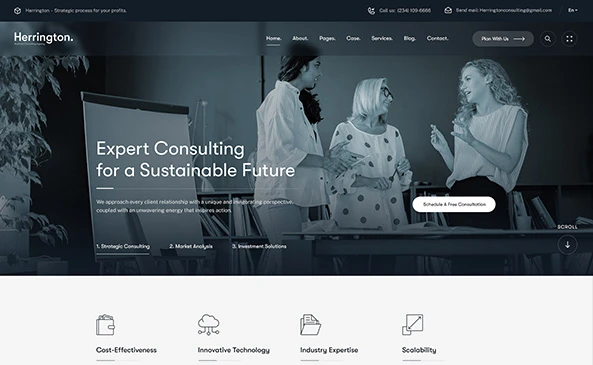
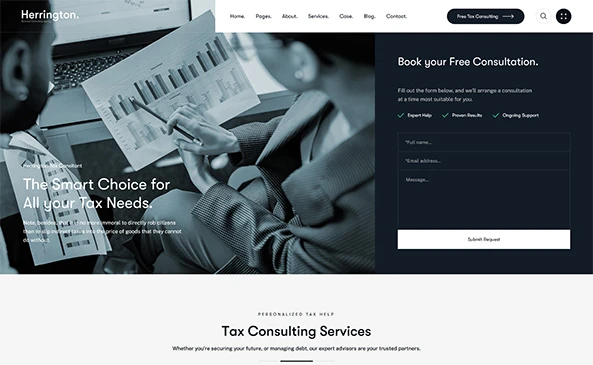
Strategic Services to Meet Global Regulatory Standards
We have vast experience in the development and approval of complex medical devices and solutions for the US FDA, EU-MDR, and global regulatory bodies.
We can help with diverse aspects of regulatory compliance, including DHF, Risk Assessment, usability, biocompatibility, safety standards compliance, cybersecurity, and AI guidance, etc.
You get regulatory deliverables as a natural result of systematic project execution and compliant documentation, which demonstrates that the product is safe, effective, and secure.


Why Vadanya Scientific ?
Seeking expert business advice early—before launching—is key. Vadanya Scientific work closely with you to identify the essential initial actions and guide your next moves.

We are a Full-Service Firm
Pre-Paid Agreements
We are a Full-Service Firm
How we Help you Limit Risks and Launch your Business.
Through our experienced MedTech consultants, Vadanya Scientific helps you manage critical early-stage tasks with precision. By handling complex processes like compliance, design, and planning, we allow you to stay focused on your broader business goals without getting lost in the technical details.

Risk Assessment & Mitigation
Regulatory Compliance Support
Launch Planning & Execution
We Develop MedTech Systems Spanning Following Technical Domains:
We provide the best services, ensuring your outstanding growth
US FDA Indian CDSCO Submissions
Design History File
ISO Standards Compliance
Risk Assessment
Safety Testing
Security and Privacy
AI Regulations
Regulatory Strategy
Let’s Collaborate with Us!
13701 Marina Pointe Dr, #210 Marina Del Rey, CA 90292
Call :+1-310-905-7597
Frequently Asked Questions
Some frequently asked questions about the service that you may have questions about